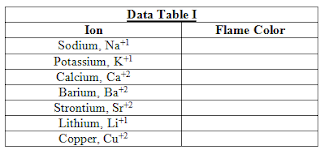
This is always such an easy lab to set up. My students like and want to do this experiment over and over again. During the expectations section of the lab, I explain to the students to carefully pay attention as each student will receive an unknown metal solution once everyone has done an initial run through.
Set-up:
Set-up:
- Make sure to purchase wooden splints (you could also use Q-Tips)
- Prepare the solutions you wish to use.
- I really recommend prepping and storing the solutions early in the school year, if at all possible.
- If you are teaching chemistry for the first time and have no experience making solutions, please order a FREE Flinn Scientific Catalog . The back of the catalog has recipes for making solutions (e.g. how much of a compound to weigh out, etc.). I do not have any affiliation with Flinn Scientific.
- Place wooden splints into each of the solutions and allow time to soak.
- The difference between the top (2010) and bottom (2015) picture is that the wooden splints were broken in half. This will allow you to save resources.
Top Picture (2010). Bottom Picture (2015).
- Place the solutions in a location that you can monitor and where access to them is open / easy.
- Instruct students - if working individually, in pairs, or groups - to test one splint per solution at a single time
- (e.g., Pair A goes and picks up a sodium-ion soaked splint. They test it and trash the splint. Pair A goes and grabs another ion soaked splint and they repeat the process until they are finished).
- Also, instruct students to properly rinse and dispose of the wooden splint (i.e., they are not to be tossed into the sink).
- Finally, if you wish, you can have an unknown beaker that contains a mixture of several of the solutions for individuals to test on their own.
- Alternatively, I have also had another set of beakers (labeled numerically or alphabetically) where students select a splint from the beaker and identify the unknown.
Of course, you could use an inoculation loop. I have also added a video (on 10/18/15) displaying the flame tests of lithium, sodium, potassium, strontium, copper (II), and an unknown.
Can you determine what the unknown is? Let me know what you think of this lab setup and/or your thoughts on the unknown solution! :)





Really Helpful thank you!
ReplyDelete